AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Pressure chemistry calculator12/26/2023 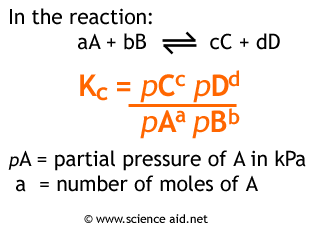
P = Pv * xA Vapour pressure of a mixture of liquids If P is the vapor pressure of the solvent, x is its mole-fraction and Pv is the vapor pressure of the pure solvent, then: The vapor pressure of liquid mixtures is calculated by using Raoult’s Law.Īccording to Raoult’s Law, the vapor pressure of a solution is directly proportional to the mole fraction of the solute. ΔHvap is the Enthalpy (heat) of Vaporization and R is the gas constant (8.3145 J mol-1 K-1). P1 and P2 are the vapor pressures at two temperatures T1 and T2. Vapor pressure is calculated by using the Clausius-Clapeyron equation as shown here: You can calculate vapour pressure using the following formulae: Some of the commonly used liquids and their vapor pressures at 20 degrees centigrade are tabulated below: Triglycerides have extremely low vapor pressures, so evaporation does not occur. Vapour pressure plays a significant role in the dynamics of cooking especially when it comes to deep-frying or cooking with fat. The boiling point of a liquid is the temperature at which vapor pressure equals atmospheric pressure.Vapour pressure also plays a critical role in calculating the time taken for evaporation of oil spills (in the case of volatile oils/chemicals).Vapour pressure is an important indicator in measuring TLV and designing safety gear for such risk-prone workers. Threshold Limit Value is a term used in assessing the safety of workers working with chemicals as a part of their daily job.It plays a significant role in classifying a substance as hazardous. 
Vapour pressure is an important property of a substance.A liquid that has more vapor pressure is said to be more volatile and evaporates faster. Vapour pressure is also the property that governs the volatility of a substance.When a liquid contains two or more immiscible liquids, irrespective of their concentration, the temperature of distillation depends on their vapor pressure. Vapor pressure is one of the factors that determine the temperature of distillation of liquids.Vapour pressure plays an important role in many daily life activities and industry applications. Note: Vapour pressure decreases with an increase in the intermolecular forces of the liquid. The following graph shows how steadily the vapor pressure of various liquids increases with temperature. This is why vapor pressure increases with an increase in temperature. When temperature increases, they attain more energy (their kinetic energy increases) and then the push becomes stronger. They keep pushing the molecules of the atmosphere with this pressure. The vapor pressure is the pressure exerted by the surface molecules of a solid/liquid in the atmosphere. I.e, when heated to a sufficient temperature, a solid turns into liquid, and a liquid converts into vapor. Thus, when sufficient heat energy is given to a substance, it is a common phenomenon that it changes its state into the next freest state possible. When heat is given to a substance, the atoms gain energy and gradually try to come out of their bonds. This is a similar concept we come across in the density of a substance too. They try to break their molecular bonds and become free. Putting it simply, the molecules of a substance always exert force on the atmosphere. Vapour pressure is the pressure exerted by a vapor when it is at equilibrium with its solid or liquid states or both, at a given temperature.Įquilibrium is a thermodynamic state of a substance where all of its possible forms (solid, liquid, gas/vapor) exist at the same time. On this online vapor pressure calculator page, you will learn what vapour pressure is, the significance of vapor pressure, and various fascinating facts about vapor pressure. Additionally, it helps in understanding the phenomenon of evaporation and volatility. Remember to check out our other calculators, for example, the osmotic pressure calculator.The vapor pressure of a substance explains its behaviour with the temperature change. Both of these equations might look intimidating at first, but, once you understand them, they turn out to be very easy. Now you know how to calculate vapor pressure on your own. When there is a big difference between the specific volume of a molecule's gas phase and its condensed phase, we can derive the following equation: It's accurate for the phase transition between liquid and gas ( vaporization) or solid and gas ( sublimation). It describes the relationship between the vapor pressure and the temperature of a liquid. The Clausius-Clapeyron equation is a derivation of this formula.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
